Authors
Saurav Roy Choudhury, Liane Babes, Jennifer J. Rahn, Bo-Young Ahn, Kimberly-Ann R. Goring, Jennifer C. King, Arthur Lau, Björn Petri, Xiaoguang Hao, Andrew K. Chojnacki, Ajitha Thanabalasuriar, Erin F. McAvoy, Sébastien Tabariès, Christoph Schraeder, Kamala D. Patel, Peter M. Siegel, Karen A. Kopciuk, David C. Schriemer, Daniel A. Muruve, Margaret M. Kelly, Bryan G. Yipp, Paul Kubes, Stephen M. Robbins, Donna L. Senger
Details
Highlights
- Dipeptidase 1 (DPEP1) serves as a vascular adhesion molecule in lungs and liver
- DPEP1 acts as a physical adhesion receptor independent of its dipeptidase activity
- Targeting DPEP1 reduces mortality in murine models of sepsis
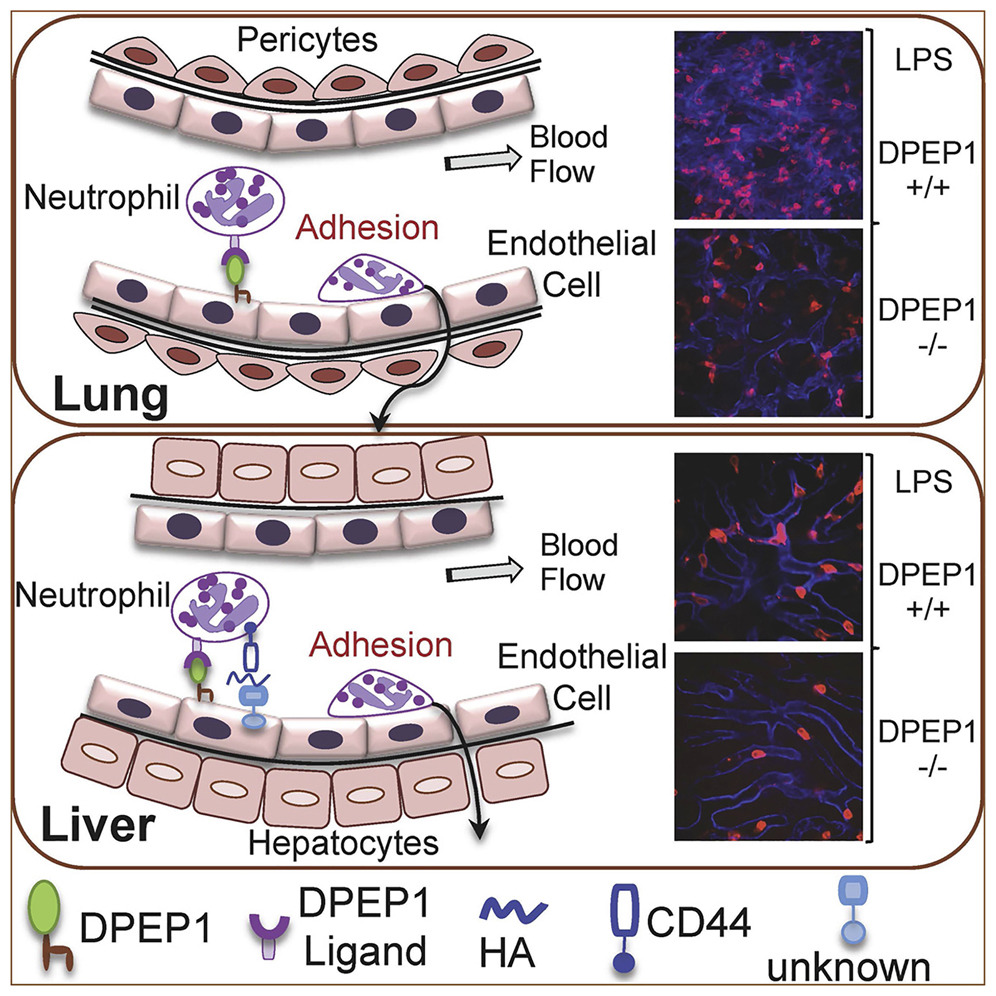
Abstract
A hallmark feature of inflammation is the orchestrated recruitment of neutrophils from the bloodstream into inflamed tissue. Although selectins and integrins mediate recruitment in many tissues, they have a minimal role in the lungs and liver. Exploiting an unbiased in vivo functional screen, we identified a lung and liver homing peptide that functionally abrogates neutrophil recruitment to these organs. Using biochemical, genetic, and confocal intravital imaging approaches, we identified dipeptidase-1 (DPEP1) as the target and established its role as a physical adhesion receptor for neutrophil sequestration independent of its enzymatic activity. Importantly, genetic ablation or functional peptide blocking of DPEP1 significantly reduced neutrophil recruitment to the lungs and liver and provided improved survival in models of endotoxemia. Our data establish DPEP1 as a major adhesion receptor on the lung and liver endothelium and identify a therapeutic target for neutrophil-driven inflammatory diseases of the lungs.